Use P.Happi® in your clinic
Join a growing network of clinicians advancing women’s intimate health
Why clinicians are joining P.Happi®:
• Supports microbiome balance without antibiotics
• Suitable for patients experiencing hormonal changes, intimate discomfort or vulvo-vaginal irritation
• Backed by Innovate UK-funded research
• Innovation powered by natural bacterium B.Y.M.® 1405 (Bdellovibrio)
Join the Clinical Network
Trusted by healthcare professionals across the UK
Get access to samples, clinical insights, and early innovation

Together, we can offer women of all life stages a safe, effective, and microbiome-friendly solution that supports comfort and confidence in daily life.
Relevant Specialists Include:
Urologists & Urogynaecologists
Gynaecologists
GPs with Women’s Health Focus
Pelvic Health Physiotherapists
Menopause Specialists
Fertility and IVF Specialists
Sexual Health Clinicians
Integrative Women’s Health Practitioners
Already used and recommended across leading clinics and specialist practices
















The Unmet Need in Recurrent Intimate Discomfort & Intimate Microbiome Imbalance
Recurrent intimate discomfort represents a substantial and ongoing clinical burden across primary and secondary care and the impact extends well beyond individual discomfort:
1.6 million women in Britain affected
Up to 70% recurrence within 12 months¹
3–10 million GP consultations annually²
£604+ million NHS cost burden³
Up to 50% of postmenopausal
Women experience symptoms associated with dryness and genitourinary changes during menopause
Ongoing Clinical Burden
Recurrent intimate health concerns remain one of the most common and persistent challenges in women’s health.
The Recurrence Cycle
Despite appropriate treatment, recurrence remains high. Many women cycle between discomfort, treatment and temporary relief without addressing underlying microbiome vulnerability.
Complementary Support
There is a growing need for microbiome-supportive strategies that complement standard care and align with stewardship principles.
P.Happi®'s microbiome appoach supports the shift toward preventative, community-based women’s healthcare, aligning with the UK Women’s Health Strategy and NHS priorities to reduce antibiotic reliance and improve long-term outcomes.
Get To Know P.Happi®
P.Happi® is a topical intimate serum designed to support the vulvo-vaginal microbiome. It contains the live beneficial bacterium Bdellovibrio bacteriovorus, which selectively targets certain disruptive bacteria associated with imbalance.
Key characteristics:
Cosmetic classification and available as self-care product in UK&I and EU (non-prescription)
Hormone-free
Topical, non-systemic
Contains live Bdellovibrio bacteriovorus
Designed for daily external application
Applied externally to the vulval area, P.Happi® supports microbiome balance without disrupting the good bacteria.

Safely Created by Women, for Women
Tested and verified by third party labs. Studies & further tests can be provided on request
Connect with our Practitioner Team
Which Patients May Benefit?
Recurrent or
Persistent Symptoms
Vaginal dryness
Microbiome imbalance
Sexual discomfort
Ongoing vulvo-vaginal irritation or imbalance
Symptoms despite negative cultures
Hormonal or Microbiome Vulnerability
Peri-/post-menopause
Postpartum or breastfeeding
Frequent antibiotic exposure
Unable or preferring to avoid local oestrogen therapy
Chronic or Sensitive Vulval Conditions
Vulval skin fragility (e.g. lichen sclerosus, alongside prescribed care)
Seeking microbiome-conscious support
Join the P.Happi® Clinical Network
As a P.Happi® Healthcare Partner, You Receive:

Product Evaluation
Access P.Happi® for evaluation, including free practitoner samples for your clinic. Opportunity to be included in our curated HCP guide.
Clinical Insights & Training
Access to webinars, case discussions and emerging research on the intimate microbiome.
Patient & Practice Resources
Educational materials and patient leaflets designed to support confident clinical conversations.
Research & Pilot Opportunities
Opportunity to participate in clinical pilots, studies or early evaluation programmes.
Tell us what you’re most interested in when you sign up, so we can tailor what we share with you.
We aim to build a collaborative, clinician-informed community focused on advancing women’s intimate health. If this sounds like you, we’d love to have you on board!
Get In Touch
Frequently Asked Questions
Have another question?
Contact Us
Is P.Happi® a treatment for UTIs?
No. P.Happi® is not a treatment and does not replace antibiotics when clinically indicated.
Can it be used alongside antibiotics?
Yes. As a topical cosmetic serum, it may be used alongside antibiotic treatment as part of a broader supportive approach.
Can it be used proactively?
P.Happi® is intended to support the natural balance of the intimate microbiome during periods of vulnerability (e.g. post-antibiotics, hormonal change, stress or sexual activity).
What is the regulatory classification?
P.Happi® is classified as a cosmetic intimate care product. It is not a medicinal product and does not claim to treat, cure or prevent disease.
Does it disrupt beneficial flora?
Bdellovibrio selectively targets Gram-negative bacteria and does not act as a broad-spectrum antibiotic.
How is this different from probiotics?
Probiotics introduce bacterial strains. Bdellovibrio works by selectively breaking down certain Gram-negative bacteria associated with imbalance.
How long does it survive?
Bdellovibrio’s survival depends on environmental conditions and the presence of susceptible bacteria. It does not colonise permanently and does not establish long-term residence in the microbiome.
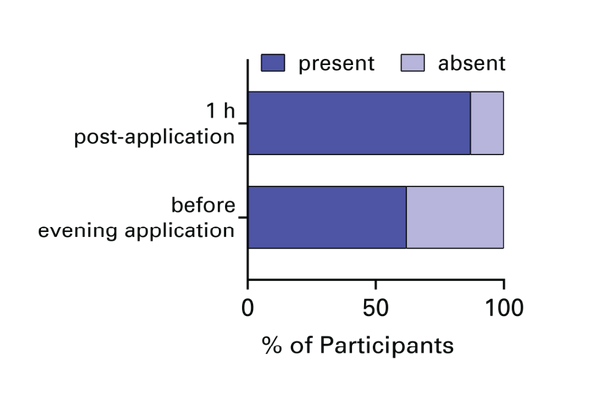
This is why we recommend applying twice daily to support consistent microbiome balance throughout the day.
How should patients use it?
P.Happi® is designed for daily external application to the vulval area and perineum as part of a regular intimate care routine. We recommend applying twice a day for optimal protection. It is non-systemic and not intended for internal use.
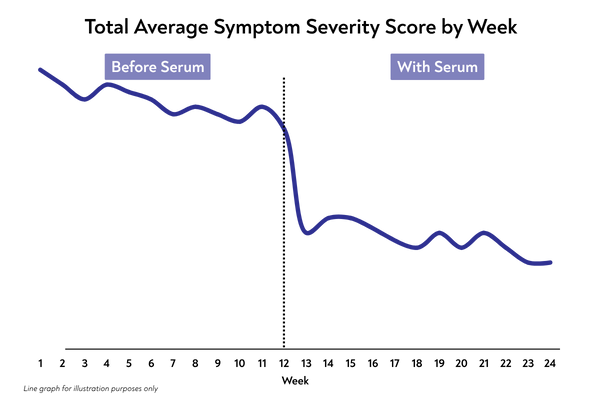
When can patients expect to feel a difference?
From first use: moisturising and soothing comfort. After ~2 weeks: many women report a growing sense of balance and support. After ~3 months: ongoing microbiome support as part of a regular routine.
Is it suitable for long-term use?
Yes. P.Happi® is designed for ongoing daily use as part of regular intimate care.
Does P.Happi® contain hormones?
No. P.Happi® is hormone-free and does not contain oestrogen, progesterone or phytoestrogens.
Can it be used in menopause?
Yes. P.Happi® is suitable for peri- and post-menopausal women.
Can it be used alongside vaginal oestrogen?
Yes. As a topical, hormone-free serum, P.Happi® may be used alongside local oestrogen therapy. It does not interfere with hormonal treatments.
Is it suitable during pregnancy or breastfeeding?
As with any intimate care product, use during pregnancy or breastfeeding should be guided by clinical judgement. P.Happi® is topical and non-systemic.
How can I recommend P.Happi® to my patients?
Healthcare professionals may recommend P.Happi® within their clinical discretion as part of a broader discussion around intimate microbiome support and as long-term management strategy.
Many practitioners choose to share a discount code that is unique to them that allows patients to purchase P.Happi® directly from our website and at a lower rate for intial purchase to trial it. This enables patients to order independently while maintaining professional separation from product distribution.
Patient information leaflets and educational materials are also available for use in clinic.
Do I need to stock P.Happi® in my clinic?
No. Most practitioners prefer to recommend P.Happi® via a practitioner code so patients can order directly online. If you are interested in in-clinic stock options for patient convenience, our team can provide further information.
Can I try P.Happi® before recommending it to patients?
Yes. Many healthcare professionals prefer to experience a product themselves before discussing it with patients.
Members of the P.Happi® Clinical Network may request a complimentary practitioner sample to review the product. Simply complete the sign-up form and our team will be in touch with details.
Do you provide materials to support patient conversations?
Yes. Healthcare partners receive access to patient information leaflets, educational resources and clinical background materials designed to support informed discussions about microbiome-conscious intimate care.
1. C.U.T.I.C. What is chronic UTI? Chronic Urinary Tract Infection Campaign. Available at: https://cutic.co.uk/what-is-chronic-uti/ 2. Health Innovation East Midlands. Digital UTI Pathway Evaluation. 2022. Available at: https://healthinnovation-em.org.uk/images/Digital_UTI_Pathway_Evaluation_-_Final_v270720.pdf 3. Public Health England. Understanding the burden of urinary tract infection hospitalisations in England. UK Health Security Agency, 2024. Available at: https://www.gov.uk/government/publications/understanding-the-burden-of-uti-hospitalisations-in-england 4. Publication pending.